This page is under construction
Drug repurposing can fill an important gap for rare disease patient groups with large unmet medical needs. In comparison to traditional drug development, drug repurposing reduces the time and costs for drug development, because safety data are mostly available. Yet, the road from initial discovery to regulatory approval and market authorization can be winding and long. We need to increase the efficiency of the drug repurposing pathway to provide broader access to new therapeutic modalities for larger groups of patients.
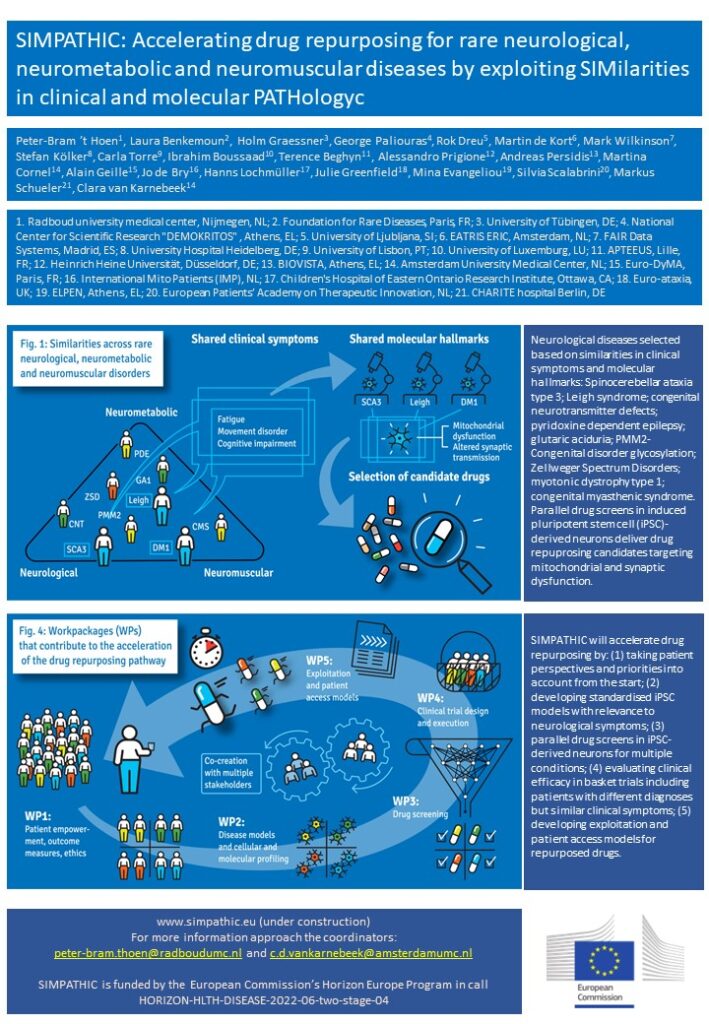
The SIMPATHIC consortium unites multidisciplinary expertise to develop a novel approach of accelerating drug repurposing for rare neurological, neurometabolic and neuromuscular disorders. The consortium, comprising patients & families, a patient training organization, pharma companies, basic, translational and clinical scientists, bio-informaticians, ethicists and methodologists, was recently awarded a HorizonEurope grant of 8.8M euros for five years. SIMPATHIC’s main innovation is the simultaneous drug development for groups of patients with different genetic diagnoses but overlapping neurological symptoms and molecular pathomechanisms.
SIMPATHIC’s key outputs accelerating the drug repurposing pathway include: Standard operating procedures for culturing stem cell-derived neuronal cell models with proven relevance for clinical symptoms and amenable to high-throughput drug screens; new drug repurposing candidates with proven efficacy in advanced brain-on-a-chip and 3D brain organoid models, as demonstrated by reversal of molecular biomarker signatures and cellular readouts associated with clinical symptoms; designs of innovative basket clinical trials to which patients with different disorders are recruited, utilizing and aggregating personalized clinical endpoints; a training module for patients and patient organizations to empower them as drivers of the drug repurposing pathway; blueprints for intellectual property strategies, business models, regulatory dossiers and patient access strategies, developed in co-creation between all relevant stakeholders.
SIMPATHIC’s proof-of-concept for the simultaneous development of repurposed drugs for multiple indications will show the path forward to development of personalized treatment opportunities for groups of rare disease patients in a cost- and time-efficient manner.
For more information, contact the coordinators: Peter-Bram ‘t Hoen (peter-bram.thoen – at – radboudumc.nl) and Clara van Karnebeek (c.d.vankarnebeek – at – amsterdamumc.nl)